On May 19, 2021 (Wednesday), we held an online webinar "Commercialization initiatives of NIH and partnership between NIH and Japanese companies (Case of Rakuten Medical Inc.)".(Host: LINK-J, Co-Host: National Institutes of Health [NIH])
Speakers
Dr. Michael L. Salgaller, Ph.D., Supervisor, Invention Development and Marketing Unit (IDMU), Technology Transfer Center | National Cancer Institute | National Institutes of Health
Dr. Miguel Garcia-Guzman, Ph.D., Founder and CSO of Rakuten Medical Inc.
Introduction of NIH's commercialization initiatives
Dr. Michael L. Salgaller, a supervisor in the Technology Transfer Center of the National Cancer Institute (NCI), gave a presentation on the commercialization initiatives of NIH.
After mentioning that there are some stereotypes that the NIH only conducts basic research and drug discovery research, and that they only collaborate with companies and academia in the U.S., he talked about how open the NIH is and the importance of worldwide partnerships with external companies and academia.
He then explained that collaboration with the NIH for the purpose of commercialization is beneficial for companies, academia and the NIH. 27 institutes and their assets/resources can provide wide-ranging support for all kinds of technologies.
While Dr. Salgaller is in charge of promoting industry-academia collaboration within NCI, 9 of the 27 institutes at NIH has outsourced promoting industry-academia collaboration to his team in NCI, and he would be happy to connect you to the other 18 institutes as well. The following is the contact information of Dr. Salgaller.
Contact: michael.salgaller@nih.gov
Lastly, he talked about the history of collaboration between NIH and Japanese companies.
NIH is actively involved in the commercialization of basic research and has conducted 85 projects with Japanese life science players so far. The ongoing collaboration between NIH and Rakuten Medical is conducting under Cooperative Research and Development Agreements.
He ended his speech by saying that he would like to continue to collaborate with Japanese companies.
He provided us with a two-page document that he would like us to share with life science players in Japan, so please have a look at it as well.
Examples of collaboration between NIH and Japanese companies (Case of Rakuten Medical Inc.)
Next, Dr. Miguel Garcia-Guzman, Founder and CSO of Rakuten Medical, who is currently collaborating with NIH, gave a lecture on the actual support and "photoimmunotherapy" technology that they are working on.
After explaining his background and company profile, he talked about the collaboration with NIH along the timeline, which started in 2011.
As for the significance of collaborating with the NIH, he concluded his presentation by describing the various support he has received, such as a support system for converting basic research into practical applications, fund procurement, smooth procedures for licensing, and continuous scientific and technical support from NIH investigators.
*The "photoimmunotherapy" technology that Rakuten Medical Inc. is currently working on for commercialization is a new cancer treatment method in which IR700 binding antibodies are injected into the patient's body, the antibodies recognize cancer cells and tumors, and the antibodies are activated by irradiating non-thermal near-infrared light to destroy the target cell membrane.

Q&A, discussion
Subsequently, Soyama from LINK-J joined the discussion.
We were able to ask some advice to companies that consider approaching the NIH and talked about the procedure for licensing the technology.
Dr. Guzman advised companies considering collaboration with the NIH. It may seem to be a daunting task, but the NIH is flexible even when it comes to early-stage technologies. Based on his experience that it was a small business at the time they started the collaboration, but received a positive response, he advised us not to hesitate to contact the NIH and they would find some way to help.

Thank you very much for your participation.
You can watch the recording of Dr. Salgaller's presentation on the LINK-J YouTube channel.







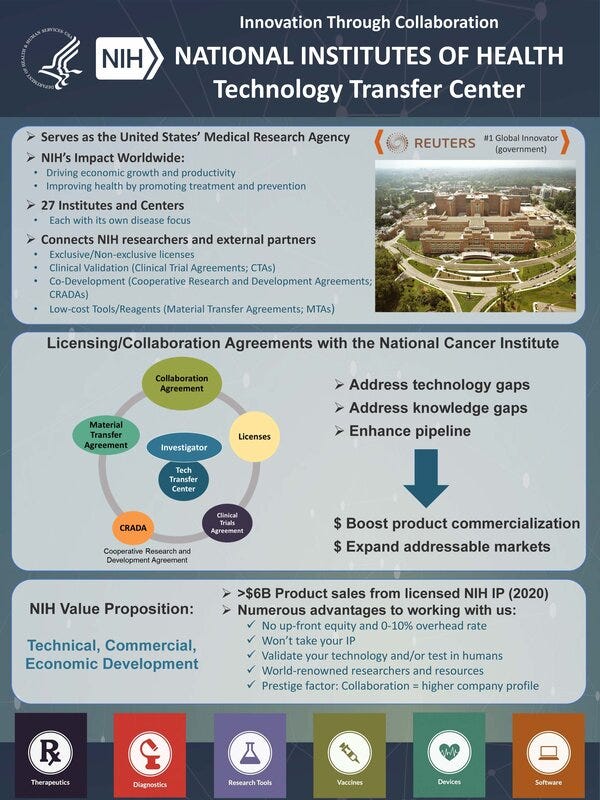 Document from Dr. Salgaller
Document from Dr. Salgaller